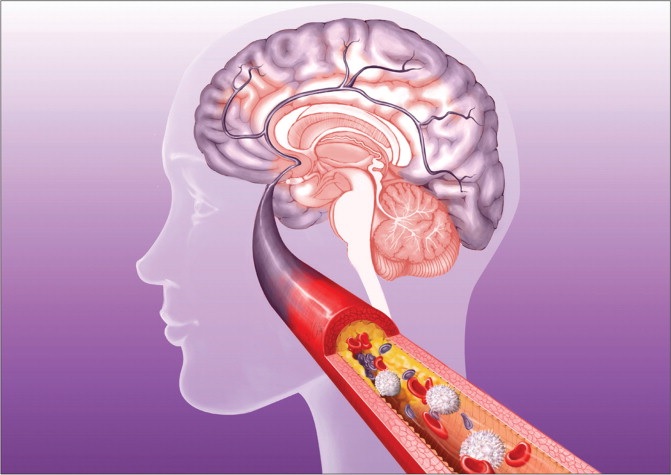
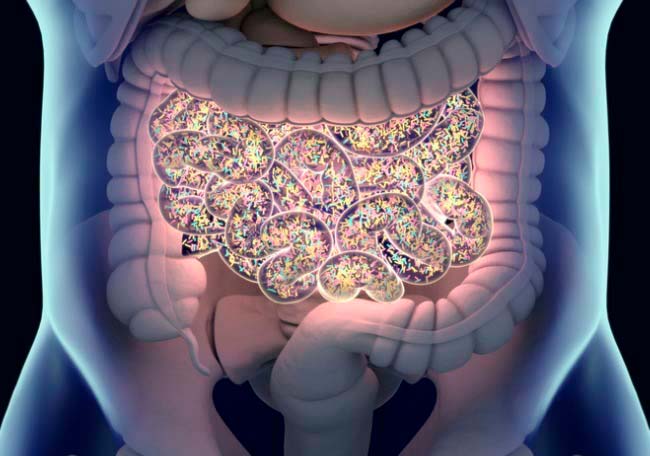
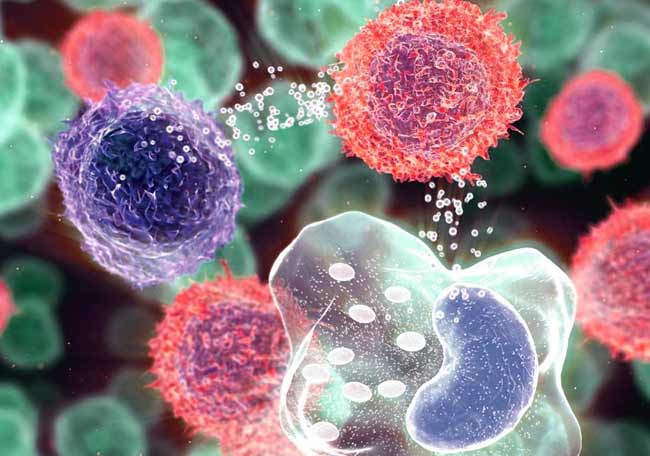
La inflamación crónica en la etiología de las enfermedades
- Furman, D. et al. Expression of specifc infammasome gene modules stratifes older individuals into two extreme clinical and immunological states. Nat. Med. 23, 174–184 (2017).
2. Netea, M. G. et al. A guiding map for infammation. Nat. Immunol. 18, 826–831 (2017).
3. Slavich, G. M. Understanding infammation, its regulation, and relevance for health: a top scientifc and public priority. Brain Behav. Immun. 45, 13–14 (2015).
4. Bennett, J. M., Reeves, G., Billman, G. E. & Sturmberg, J. P. Infammation– nature’s way to efciently respond to all types of challenges: implications for understanding and managing “the epidemic” of chronic diseases. Front. Med. 5, 316 (2018).
5. GBD 2017 Causes of Death Collaborators. Global, regional, and national age-sex-specifc mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392, 1736–1788 (2018).
6. Miller, G. E., Chen, E. & Parker, K. J. Psychological stress in childhood and susceptibility to the chronic diseases of aging: moving toward a model of behavioral and biological mechanisms. Psychol. Bull. 137, 959–997 (2011).
7. Fleming, T. P. et al. Origins of lifetime health around the time of conception: causes and consequences. Lancet 391, 1842–1852 (2018).
8. Renz, H. etal. An exposome perspective: early-life events and immune development in a changing world. J. Allergy Clin. Immunol. 140, 24–40 (2017).
9. Kotas, M. E. & Medzhitov, R. Homeostasis, infammation, and disease susceptibility. Cell 160, 816–827 (2015).
10. Straub, R. H., Cutolo, M., Buttgereit, F. & Pongratz, G. Energy regulation and neuroendocrine-immune control in chronic infammatory diseases. J. Intern. Med. 267, 543–560 (2010).
11. Straub, R. H., Cutolo, M. & Pacifci, R. Evolutionary medicine and bone loss in chronic infammatory diseases—a theory of infammation-related osteopenia. Semin. Arthritis Rheum. 45, 220–228 (2015).
12. Straub, R. H. & Schradin, C. Chronic infammatory systemic diseases: an evolutionary trade-of between acutely benefcial but chronically harmful programs. Evol. Med. Public Health 2016, 37–51 (2016).
13. Straub, R. H. Te brain and immune system prompt energy shortage in chronic infammation and ageing. Nat. Rev. Rheumatol. 13, 743–751 (2017).
14. Slavich, G. M. Psychoneuroimmunology of stress and mental health. in Te Oxford Handbook of Stress and Mental Health (eds K. Harkness & E. P. Hayden) (Oxford University Press, in the press).
15. Fullerton, J. N. & Gilroy, D. W. Resolution of infammation: a new therapeutic frontier. Nat. Rev. Drug Discov. 15, 551–567 (2016).
16. Calder, P. C. et al. A consideration of biomarkers to be used for evaluation of infammation in human nutritional studies. Br. J. Nutr. 109, S1–S34 (2013).
17. Taniguchi, K. & Karin, M. NF-κB, infammation, immunity and cancer: coming of age. Nat. Rev. Immunol. 18, 309–324 (2018).
18. Gisterå, A. & Hansson, G. K. Te immunology of atherosclerosis. Nat Rev. Nephrol. 13, 368–380 (2017).
19. Ferrucci, L. & Fabbri, E. Infammageing: chronic infammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 15, 505–522 (2018).
20. Heneka, M. T., Kummer, M. P. & Latz, E. Innate immune activation in neurodegenerative disease. Nat. Rev. Immunol. 14, 463–477 (2014).
21. Miller, A. H. & Raison, C. L. Te role of infammation in depression: from evolutionary imperative to modern treatment target. Nat. Rev. Immunol. 16, 22–34 (2016).
22. Shen-Orr, S. S. et al. Defective signaling in the JAK-STAT pathway tracks with chronic infammation and cardiovascular risk in aging humans. Cell Syst. 3, 374–384.e4 (2016).
23. Verschoor, C. P. et al. Serum C-reactive protein and congestive heart failure as signifcant predictors of herpes zoster vaccine response in elderly nursing home residents. J. Infect. Dis. 216, 191–197 (2017).
24. Fourati, S. et al. Pre-vaccination infammation and B-cell signalling predict age-related hyporesponse to hepatitis B vaccination. Nat. Commun. 7, 10369 (2016).
25. McDade, T. W., Adair, L., Feranil, A. B. & Kuzawa, C. Positive antibody response to vaccination in adolescence predicts lower C-reactive protein concentration in young adulthood in the Philippines. Am. J. Hum. Biol. 23, 313–318 (2011).
26. Singer, K. & Lumeng, C. N. Te initiation of metabolic infammation in childhood obesity. J. Clin. Invest. 127, 65–73 (2017).
27. Olvera Alvarez, H. A., Kubzansky, L. D., Campen, M. J. & Slavich, G. M. Early life stress, air pollution, infammation, and disease: an integrative review and immunologic model of social-environmental adversity and lifespan health. Neurosci. Biobehav. Rev. 92, 226–242 (2018).
28. Serhan, C. N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 510, 92–101 (2014).
29. Serhan, C. N. & Levy, B. D. Resolvins in infammation: emergence of the pro-resolving superfamily of mediators. J. Clin. Invest. 128, 2657–2669 (2018).
30. Franceschi, C., Garagnani, P., Vitale, G., Capri, M. & Salvioli, S. Infammaging and ‘garb-aging’. Trends Endocrinol. Metab. 28, 199–212 (2017).
31. Liston, A. & Masters, S. L. Homeostasis-altering molecular processes as mechanisms of infammasome activation. Nat. Rev. Immunol. 17, 208–214 (2017).
32. Frank, D. & Vince, J. E. Pyroptosis versus necroptosis: similarities, diferences, and crosstalk. Cell Death Difer. 26, 99–114 (2019).
33. Jin, C., Henao-Mejia, J. & Flavell, R. A. Innate immune receptors: key regulators of metabolic disease progression. Cell Metab. 17, 873–882 (2013).
34. Hotamisligil, G. S. Infammation, metafammation and immunometabolic disorders. Nature 542, 177–185 (2017).
35. Kazankov, K. et al. Te role of macrophages in nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Nat. Rev. Gastroenterol. Hepatol. 16, 145–159 (2019).
36. Redlich, K. & Smolen, J. S. Infammatory bone loss: pathogenesis and therapeutic intervention. Nat. Rev. Drug Discov. 11, 234–250 (2012).
37. Zhang, J. et al. Te risk of metabolic syndrome in patients with rheumatoid arthritis: a meta-analysis of observational studies. PLoS One 8, e78151 (2013).
38. Armstrong, A. W., Harskamp, C. T. & Armstrong, E. J. Psoriasis and the risk of diabetes mellitus: a systematic review and meta-analysis. JAMA Dermatol. 149, 84–91 (2013).
39. Dregan, A., Charlton, J., Chowienczyk, P. & Gulliford, M. C. Chronic infammatory disorders and risk of type 2 diabetes mellitus, coronary heart disease, and stroke: a population-based cohort study. Circulation 130, 837–844 (2014).
40. Ridker, P. M. A test in context: high-sensitivity C-reactive protein. J. Am. Coll. Cardiol. 67, 712–723 (2016).
41. Emerging Risk Factors Collaboration. et al. C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: an individual participant meta-analysis. Lancet 375, 132–140 (2010).
42. Burska, A. N., Sakthiswary, R. & Sattar, N. Efects of tumour necrosis factor antagonists on insulin sensitivity/resistance in rheumatoid arthritis: a systematic review and meta-analysis. PLoS One 10, e0128889 (2015).
43. Chou, R. et al. Treatment for rheumatoid arthritis and risk of Alzheimer’s disease: a nested case-control analysis. CNS Drugs 30, 1111–1120 (2016).
44. Ridker, P. M. et al. Antiinfammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017).
45. Proctor, M. J. et al. Systemic infammation predicts all-cause mortality: a Glasgow infammation outcome study. PLoS One 10, e0116206 (2015).
46. Arai, Y. et al. Infammation, but not telomere length, predicts successful ageing at extreme old age: a longitudinal study of semi-supercentenarians. EBioMedicine 2, 1549–1558 (2015).
47. Roubenof, R. et al. Monocyte cytokine production in an elderly population: efect of age and infammation. J. Gerontol. A Biol. Sci. Med. Sci. 53, M20–M26 (1998).
48. Ahluwalia, N. et al. Cytokine production by stimulated mononuclear cells did not change with aging in apparently healthy, well-nourished women. Mech. Ageing Dev. 122, 1269–1279 (2001).
49. Beharka, A. A. et al. Interleukin-6 production does not increase with age. J. Gerontol. A Biol. Sci. Med. Sci. 56, B81–B8 (2001).
50. Elisia, I. et al. Efect of age on chronic infammation and responsiveness to bacterial and viral challenges. PLoS One 12, e0188881 (2017).
51. Morrisette-Tomas, V. et al. Infamm-aging does not simply refect increases in pro-infammatory markers. Mech. Ageing Dev. 139, 49–57 (2014). 52. Alpert, A. et al. A clinically meaningful metric of immune age derived from high-dimensional longitudinal monitoring. Nat. Med. 25, 487–495 (2019).
53. Coppé, J.-P., Desprez, P.-Y., Krtolica, A. & Campisi, J. Te senescenceassociated secretory phenotype: the dark side of tumor suppression. Annu. Rev. Pathol. 5, 99–118 (2010).
54. Zhu, Y., Armstrong, J. L., Tchkonia, T. & Kirkland, J. L. Cellular senescence and the senescent secretory phenotype in age-related chronic diseases. Curr. Opin. Clin. Nutr. Metab. Care 17, 324–328 (2014).
55. Kennedy, B. K. et al. Geroscience: linking aging to chronic disease. Cell 159, 709–713 (2014).
56. Campisi, J. Aging, cellular senescence, and cancer. Annu. Rev. Physiol. 75, 685–705 (2013).
57. Efros, R. B. Te silent war of CMV in aging and HIV infection. Mech. Ageing Dev. 158, 46–52 (2016).
58. Stout, M. B., Justice, J. N., Nicklas, B. J. & Kirkland, J. L. Physiological aging: links among adipose tissue dysfunction, diabetes, and frailty. Physiology (Bethesda) 32, 9–19 (2017).
59. Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. & Santoro, A. Infammaging: a new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 159, 1–15 (2018). 60. Zitvogel, L., Pietrocola, F. & Kroemer, G. Nutrition, infammation and cancer. Nat. Immunol. 18, 843–850 (2017).
61. Razzoli, M. et al. Social stress shortens lifespan in mice. Aging Cell 17, e12778 (2018).
62. Carroll, J. E. et al. Partial sleep deprivation activates the DNA damage response (DDR) and the senescence-associated secretory phenotype (SASP) in aged adult humans. Brain Behav. Immun. 51, 223–229 (2016).
63. Yuan, J. et al. Long-term persistent organic pollutants exposure induced telomere dysfunction and senescence-associated secretary phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 73, 1027–1035 (2018).
64. Shen-Orr, S. S. & Furman, D. Variability in the immune system: of vaccine responses and immune states. Curr. Opin. Immunol. 25, 542–547 (2013).
65. McDade, T. W. Early environments and the ecology of infammation. Proc. Natl Acad. Sci. USA 109, 17281–17288 (2012).
66. Carrera-Bastos, P., Fontes-Villalba, M., O’Keefe, J. H., Lindeberg, S. & Cordain, L. Te western diet and lifestyle and diseases of civilization. Res. Rep. Clin. Cardiol. 2, 15–35 (2011).
67. Raichlen, D. A. et al. Physical activity patterns and biomarkers of cardiovascular disease risk in hunter-gatherers. Am. J. Hum. Biol. 29, e22919 (2017).
68. Kaplan, H. et al. Coronary atherosclerosis in indigenous South American Tsimane: a cross-sectional cohort study. Lancet 389, 1730–1739 (2017).
69. Lindeberg, S. & Lundh, B. Apparent absence of stroke and ischaemic heart disease in a traditional Melanesian island: a clinical study in Kitava. J. Intern. Med. 233, 269–275 (1993).
70. Lindeberg, S., Berntorp, E., Nilsson-Ehle, P., Terént, A. & Vessby, B. Age relations of cardiovascular risk factors in a traditional Melanesian society: the Kitava Study. Am. J. Clin. Nutr. 66, 845–852 (1997).
71. Lindeberg, S., Eliasson, M., Lindahl, B. & Ahrén, B. Low serum insulin in traditional Pacifc Islanders—the Kitava Study. Metabolism 48, 1216–1219 (1999).
72. Brodin, P. et al. Variation in the human immune system is largely driven by non-heritable infuences. Cell 160, 37–47 (2015).
73. Niedzwiecki, M. M. et al. Te exposome: molecules to populations. Annu. Rev. Pharmacol. Toxicol. 59, 107–127 (2019).
74. Virgin, H. W., Wherry, E. J. & Ahmed, R. Redefning chronic viral infection. Cell 138, 30–50 (2009).
75. Wang, C. et al. Efects of aging, cytomegalovirus infection, and EBV infection on human B cell repertoires. J. Immunol. 192, 603–611 (2014).
76. Petta, S. et al. Hepatitis C virus infection is associated with increased cardiovascular mortality: a meta-analysis of observational studies. Gastroenterology 150, 145–155.e4 (2016).
77. Root-Bernstein, R. & Fairweather, D. Complexities in the relationship between infection and autoimmunity. Curr. Allergy Asthma Rep. 14, 407 (2014).
78. Furman, D. et al. Cytomegalovirus infection enhances the immune response to infuenza. Sci. Transl. Med. 7, 281ra43 (2015).
79. Pawelec, G. et al. Human immunosenescence: is it infectious? Immunol. Rev. 205, 257–268 (2005).
80. Chou, J. P., Ramirez, C. M., Wu, J. E. & Efros, R. B. Accelerated aging in HIV/AIDS: novel biomarkers of senescent human CD8+ T cells. PLoS One 8, e64702 (2013).
81. Sochocka, M., Zwolińska, K. & Leszek, J. Te infectious etiology of Alzheimer’s disease. Curr. Neuropharmacol. 15, 996–1009 (2017).
82. Rook, G., Bäckhed, F., Levin, B. R., McFall-Ngai, M. J. & McLean, A. R. Evolution, human-microbe interactions, and life history plasticity. Lancet 390, 521–530 (2017).
83. McDade, T. W. et al. Analysis of variability of high sensitivity C-reactive protein in lowland Ecuador reveals no evidence of chronic low-grade infammation. Am. J. Hum. Biol. 24, 675–681 (2012).
84. Liebert, M. A. et al. Implications of market integration for cardiovascular and metabolic health among an indigenous Amazonian Ecuadorian population. Ann. Hum. Biol. 40, 228–242 (2013).
85. Eriksson, U. K., van Bodegom, D., May, L., Boef, A. G. C. & Westendorp, R. G. J. Low C-reactive protein levels in a traditional West-African population living in a malaria endemic area. PLoS One 8, e70076 (2013).
86. Agmon-Levin, N. et al. Antitreponemal antibodies leading to autoantibody production and protection from atherosclerosis in Kitavans from Papua New Guinea. Ann. N. Y. Acad. Sci. 1173, 675–682 (2009).
87. Gurven, M., Jaeggi, A. V., Kaplan, H. & Cummings, D. Physical activity and modernization among Bolivian Amerindians. PLoS One 8, e55679 (2013).
88. Cordain, L. et al. Plant-animal subsistence ratios and macronutrient energy estimations in worldwide hunter-gatherer diets. Am. J. Clin. Nutr. 71, 682–692 (2000).
89. Kuipers, R. S., Joordens, J. C. A. & Muskiet, F. A. J. A multidisciplinary reconstruction of Palaeolithic nutrition that holds promise for the prevention and treatment of diseases of civilisation. Nutr. Res. Rev. 25, 96–129 (2012).
90. De la Iglesia, H. O. et al. Ancestral sleep. Curr. Biol. 26, R271–R272 (2016). 91. Slavich, G. M. & Cole, S. W. Te emerging feld of human social genomics. Clin. Psychol. Sci. 1, 331–348 (2013).
92. Chakravarthy, M. V. & Booth, F. W. Eating, exercise, and ‘thrify’ genotypes: connecting the dots toward an evolutionary understanding of modern chronic diseases. J. Appl. Physiol. 96, 3–10 (2004).
93. Hallal, P. C. et al. Global physical activity levels: surveillance progress, pitfalls, and prospects. Lancet 380, 247–257 (2012).
Comentarios
Para ver los comentarios de sus colegas o para expresar su opinión debe ingresar con su cuenta de IntraMed.